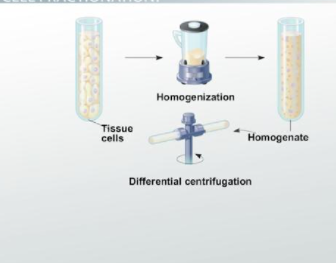
Have you ever wondered how scientists discovered that mitochondria are the cell’s powerhouses or how they found out that the endoplasmic reticulum functions differently from the Golgi apparatus? By isolating organelles in a technique known as cell fractionation, scientists were able to determine their activities.
In a nutshell, Cell fractionation is the process of breaking down cells and separating the many organelles they contain. It’s utilized to aid in the investigation of cell architecture and functions. Homogenization and ultracentrifugation are the two stages of cell fractionation.
A cell is divided into its working organelles and macromolecules to divide the tissue into its live constituent cell types. Although biochemical research necessitates destroying the cell’s anatomy, gentle fractionation procedures separate the distinct cell components while maintaining their functions.
Cells can be split apart by various methods, including osmotic shock or ultrasonic vibration, forcing through a small hole, or blending. Many of the cell’s membranes (including the plasma membrane and endoplasmic reticulum membranes) break into fragments during these procedures, which then reseal to create small, closed vesicles. Disruption techniques leave nuclei, mitochondria, the Golgi apparatus, lysosomes, and peroxisomes generally intact when used judiciously. The cell suspension converts to a thick slurry (referred to as a homogenate or extract) containing various membrane-enclosed organelles, each with its size, charge, and density.
The numerous components, including the vesicles produced from the endoplasmic reticulum, called microsomes, retain most of their original biochemical features if the homogenization medium has been carefully chosen (by trial and error for each organelle).
Separate the homogenate components next. Only with the commercial introduction of the preparative ultracentrifuge in the early 1940s, extracts of shattered cells rotate at high speeds, making such cell fractionations possible. This therapy divides cell components based on their size and density: the larger units, in general, are subjected to the most significant centrifugal force and move the fastest.
Large parts such as nuclei settle to the bottom of the centrifuge tube to form a pellet; a pellet of mitochondria drops at a little higher speed; and at even higher rates and for more extended periods of centrifugation, small, closed vesicles and then recover ribosomes. Although all of these fractions are contaminated, many impurities are eliminated by resuspending the pellet and centrifuging it multiple times.
Place the tissue in a cold, isotonic buffer solution. This solution reduces the enzyme activity that breaks down organelles, and because the buffer is isotonic, it prevents the organelles from expanding and bursting or shrinking. Additionally, the buffer maintains a constant pH so that the cell does not denature or become damaged in any manner during the process of fractionation.
Differential centrifugation is the term used to describe the entire process, which involves centrifuging at different speeds. There is a relationship between each organelle’s function and its structure, the same way the structure of proteins determines their function. As a result, spinning the solution at appropriate speeds can separate organelles from one another. If you span the solution at a given speed for a specific amount of time, you will be able to separate your specific organelle for further investigation and analysis.
A short history and the primary objective of Cell Fractionation
Albert Claude (1899-1983), a pioneer in cell separation by differential centrifugation, worked in the 1930s and 1940s. He was the first to utilize electron microscopes to examine cells when they became accessible in the early 1940s. Revolutionized perceptions of these scientists were due to these combined approaches: what had appeared to be watery and formless was found to have an internal organization that allows the study of its structure, biochemistry, and function.
This “maturing,” as Claude described it, was heralded by Claude and his colleagues. Using an electron microscope, they created the first image of an entire cell. Claude also found the lace-like endoplasmic reticulum and recognized mitochondria as the cell’s powerhouses, rich in enzymes responsible for oxygen uptake. For their “discoveries concerning the functional and structural organization of the cell,” he shared the Nobel Prize in 1974 with Christian de Duve (1917- ) and George E. Palade (1912-2008).
In 1929, Claude joined James B. Murphy’s Rockefeller cancer laboratory to isolate the Rous sarcoma virus using scientific methods. Scientists were still debating whether Peyton Rous’s tumor-causing agent, discovered in 1911 at Rockefeller, was a virus at the time. Claude carefully broke open infected cells with a mortar and pestle to isolate the agent, then centrifuged the contents to separate sub-cellular components by density into “fractions” that he then tested.
Claude worked on improving the technique for numerous years. He separated mitochondria and discovered particles that became known as microsomes while extracting and purifying the Rous agent. This action was the first electron micrograph to reveal an intact cell. The image was provided by the Rockefeller Archive Center.
In 1943, Claude published an article in Science that led to a cooperation with Ernest Fullam at the Interchemical Laboratory in New York, which had one of the country’s few electron microscopes. Meanwhile, Keith Porter (1912-1997) had joined Claude’s research group and devised a technique for growing cells (chicken embryo fibroblasts) thin enough to be observed with an electron microscope. Their collaboration resulted in the first electron microscopy image of an intact cell in 1945, in an attempt to see where microsomes and other structures in the cell were situated. Please later dubbed this image the “birth certificate” of the discipline of cell biology since it was so rich in structure and information.
The following process briefly summarizes the Cell Fractionation process
- Slice the tissue into pieces and place them in an isotonic buffering solution (in cold conditions).
- To release the organelles from the cell, you can use a blender to homogenize the tissue.
- After the formation of a fluid known as homogenate, filter to separate the homogenized suspension.
- Centrifuge the filtrate at a low speed.
- Decant the supernatant and re-centrifuge it at a higher speed until the desired organelle is separated.
- Analyze the contents of the supernatant.
What are the functions of Cell Fractionation?
- Protein Enrichment: Enhance the identification of low-abundance proteins by enriching target proteins.
- Protein Characterization: Determine how proteins distribute themselves within the cell.
- Protein Translocation: Keep track of cell signaling molecules as they move from the cytoplasm to the nucleus.
Advantages of Cell Fractionation
- The technique’s main advantages are that it’s quick and easy to do, and it usually only requires a high-speed centrifuge, which is common in laboratories.
- Usable together with other separation techniques.
Disadvantages of Cell Fractionation:
- The technique’s main drawback is that it only separates cellular components with significant size differences. Therefore, the fractions are normally crude.
- Cell Fractionation requires specialized instrumentation.